Why Are Eye Drop Caps So Hard to Open? The Design Problem Nobody Talks About
For millions of patients, it's the biggest obstacle in their day.
You've been there. You need your eye drops — maybe it's first thing in the morning, maybe it's after a long day at a screen, maybe it's the middle of the night after surgery. The bottle is right there in your hand. And the cap won't budge.
You press down. You twist. You press harder. You try again with a dish towel for grip. Maybe you run it under hot water. At some point, frustration gives way to something worse — pliers, scissors, or your teeth. And somewhere in the back of your mind, a quiet but real worry surfaces: what if I can't do this myself?
This is a story that plays out millions of times a day, in bathrooms and bedrooms and hospital recovery rooms around the world. And almost nobody in the pharmaceutical industry is talking about it.
The Bottle Was Never Really Designed for You
Here's the uncomfortable truth at the center of this problem: the standard eye drop bottle was designed primarily around two competing priorities — keeping children out and keeping the sterile product in. The patient using it daily, often with impaired vision, reduced grip strength, or shaky hands, was largely an afterthought.
Child-resistant (CR) packaging regulations, governed in the U.S. by the Poison Prevention Packaging Act and enforced by the Consumer Product Safety Commission, require caps to resist opening by at least 85% of children under five. That's a worthy goal. But there is a design tradeoff built into this standard that the industry rarely acknowledges openly: a cap engineered to defeat a determined five-year-old will also, reliably and predictably, defeat an elderly glaucoma patient with arthritic hands.
“…a cap engineered to defeat a determined five-year-old will also, reliably and predictably, defeat an elderly glaucoma patient with arthritic hands."
Industrial designers and human factors engineers have a name for this phenomenon: the Usability Gap. It describes the measurable disconnect between what a regulatory standard demands and what the actual end-user can physically accomplish. In the case of CR eye drop caps, the gap is not theoretical — it is physiological. Opening a standard 17–19mm push-and-turn cap requires meaningful pinch force and torque. And research published through the National Institutes of Health reveals that pinch strength — the precise grip mechanic required to depress and twist a small bottle cap — declines by approximately 23% in males and 27% in females by age 80. The "standard adult" assumed by CR testing protocols bears little resemblance to the patient who actually fills that prescription.
This failure is also one that the field of Universal Design has long anticipated. Universal Design — the framework for creating products usable by all people without need for adaptation — includes a core principle that speaks directly to this issue: Principle 6: Low Physical Effort, which holds that a design should be usable efficiently and comfortably with minimum fatigue. The standard eye drop cap fails this principle categorically. It demands torque and pinch force that a significant portion of its user base simply cannot generate — not due to carelessness or inattention, but due to the normal physical realities of aging and illness.
Eye drop bottles are also significantly smaller than most other medication containers, which compounds the problem. As researchers at Pharmaceutical Technology have noted, the limited surface area available for gripping makes extra effort almost unavoidable when opening the cap — and that effort becomes exponentially harder for anyone already dealing with reduced dexterity.
Who's Really Struggling — And How Many of Them Are There?
This is not a niche problem affecting a small slice of the population. The overlap between the conditions that make eye drops necessary and the conditions that make caps hard to open is vast — and it is growing faster than most people realize.
"The system of medication use is a complex one, and each step is prone to error."
– Lucian Leape, M.D. (To Err Is Human: Building a Safer Health System)
Start with arthritis. According to the Centers for Disease Control and Prevention, approximately 47.3% of U.S. adults aged 65 and older have doctor-diagnosed arthritis — a leading cause of reduced manual dexterity and diminished grip strength. Nearly half of the primary demographic for prescription eye drops is contending with the exact physical limitation that makes standard caps so difficult to open. This is not an edge case. It is the statistical norm.
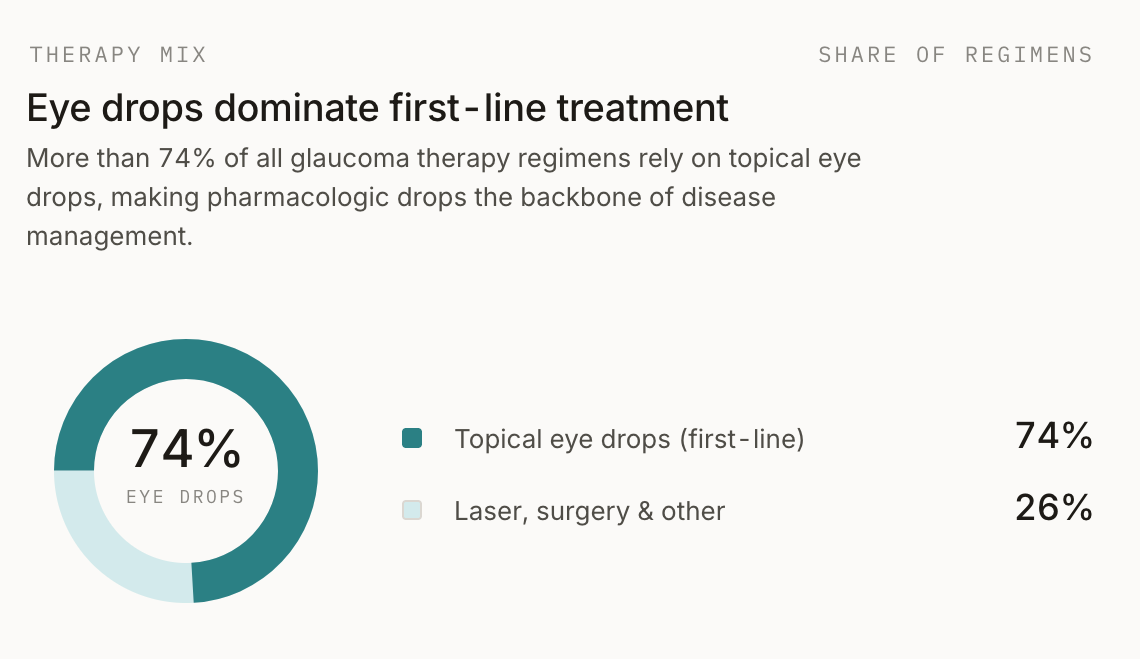
Glaucoma alone affects over 79 million people worldwide as of 2024, with open-angle glaucoma — the form most commonly managed with daily eye drops — accounting for approximately 63 million of those cases. The global glaucoma treatment market was valued at nearly $9 billion in 2024 and is projected to reach over $14 billion by 2034. Eye drops remain the first-line treatment, accounting for more than 74% of all glaucoma therapy regimens.
Glaucoma is, critically, a disease of aging. The typical glaucoma patient is an older adult, and a peer-reviewed study found that 62% of people attending a rheumatoid arthritis clinic reported difficulty instilling eye drops — making them four-and-a-half times more likely to experience difficulty than patients in a general ophthalmology clinic. More broadly, researchers estimate that as many as 50% of all patients face physical difficulties administering eye drops, yet many are unaware of, or reluctant to admit, these difficulties to their doctors.
And the population at the intersection of all these challenges is not shrinking. The World Health Organization projects that by 2050, the global population of people aged 60 and older will double to 2.1 billion. The patients most vulnerable to packaging that ignores their physical realities are not a passing demographic. They are the future of healthcare — and the fastest-growing segment of the prescription eye drop market.
The Hidden Cost: Missed Doses, Compromised Treatment, and a Psychological Spiral
If this were merely an inconvenience, it might be easier to overlook. But the stakes of not opening an eye drop bottle correctly — or not opening it at all — are severe, in ways that extend from the individual patient all the way to the healthcare system.
Medication adherence for glaucoma is already a significant clinical problem. Only 20% to 70% of glaucoma patients use their medication as prescribed, and roughly half discontinue treatment within the first six months. The physical difficulty of managing the bottle is consistently cited as one of the primary reasons. For glaucoma patients specifically, poor adherence isn't just inconvenient — it can mean progressive, irreversible vision loss and escalation to more aggressive surgical intervention.'
"Only 20% to 70% of glaucoma patients use their medication as prescribed…"
– Patient Challenges with Glaucoma Eye Drops — Ophthalmology Glaucoma (2024)
The economic consequences of this broader non-adherence crisis are staggering. Across all medications, the National Association of Boards of Pharmacy estimates that medication non-adherence costs the U.S. healthcare system between $100 billion and $300 billion annually in avoidable medical costs — through increased hospitalizations, emergency visits, and disease complications that proper medication management could have prevented. Every patient who cannot open their bottle is a small tributary feeding that enormous downstream cost.
There is also a psychological dimension to this problem that the clinical literature has begun to examine more closely. Behavioral psychologist Albert Bandura's Self-Efficacy Theory offers a compelling lens: a person's belief in their own ability to complete a task directly predicts whether they will continue attempting it. When a patient repeatedly fails to open their medication — pressing, twisting, struggling — their task-specific self-efficacy erodes. The frustration is not just physical. It is a quiet, cumulative message that this is something they cannot do. Over time, that belief becomes the real barrier to adherence, long after the physical struggle has faded from memory.
The workarounds patients improvise when they give up on proper opening aren't just ineffective — they can be dangerous. Medical researchers have documented patients using knives and scissors to puncture bottle tips, creating risks of both injury and contamination. One clinical commentator noted that ophthalmologists often don't assess whether patients are actually able to successfully instill their drops in the first place — and may prescribe "without thinking about the bottle."
Even the FDA has taken notice. The agency's MedWatch system actively monitors complaints about eye drop packaging, and in at least one notable case, the FDA required a manufacturer to revise its torque specifications after post-marketing studies confirmed that patients were physically unable to open the bottles as packaged.
The Design Gap Nobody Has Solved
What makes this problem remarkable is not that it exists — it's that it has persisted, at massive scale, for so long without a mainstream solution.
The pharmaceutical packaging industry has made genuine advances in tamper-evident seals, contamination prevention, and sterility assurance. The standard 2.4mL eye drop bottle — with its 17–19mm cap, its tamper-evident ring, and its push-and-turn mechanism — remains largely unchanged. Tens of millions of patients encounter it every day.
The field of Universal Design has offered a clear framework for what better packaging should look like for decades. The gap is not a lack of knowledge about what good design requires. It is a lack of prioritization — a failure to treat the patient's daily, physical experience of the product as a design constraint worthy of the same rigor applied to sterility or tamper-resistance.
What a Real Solution Looks Like
The Eye Drop Bottle Opener Set from Useful & Usable Workshop is a direct answer to this gap — built not around regulatory compliance, but around the actual, physiological reality of the people who depend on these bottles every day.
Designed in Columbus, Ohio, this two-piece system — a Comfort Grip Handle for stabilizing the bottle and a Gentle Turn Wrench for removing the cap — addresses the Usability Gap directly. The Comfort Grip Handle eliminates the challenge of holding a small, slippery bottle steady. The Gentle Turn Wrench uses a patent-pending compliant mechanism that adapts to the bottle cap with a simple press, gripping it securely without requiring the pinch force and torque that decline so significantly with age.
In the language of Universal Design, it embodies Principle 6 in practice: low physical effort, minimum fatigue, accessible to the broadest possible range of users. In the language of Self-Efficacy Theory, it gives patients back something more important than an open bottle — it restores their belief that they can manage their own care, reliably, day after day.
It's not a workaround. It's a purpose-built tool for a problem that has been quantified, documented, and under-addressed for far too long.
The Bigger Picture
Every day that a patient struggles with their eye drop cap, something small but significant erodes: their confidence in managing their own health, their sense of independence, and ultimately their adherence to a treatment that may be protecting their vision.
The numbers behind this problem are not abstract. Nearly half of seniors have arthritis. Fifty percent of eye drop patients struggle with administration. The aging global population is doubling. Non-adherence is costing the healthcare system billions of dollars annually. And the cap on the standard eye drop bottle has not materially changed.
The design problem is real, it is documented, and it is consequential. The good news is that it is also solvable — not by redesigning the entire pharmaceutical supply chain, but by meeting patients where they are, with tools designed around how they actually live and what their hands can actually do.
That's the gap. And that's where innovation belongs.
The Eye Drop Bottle Opener Set is available at uuworkshop.com and on Etsy. Disclaimer: This product is intended as a convenience aid and is not a medical device. It is not intended to diagnose, treat, cure, mitigate, or prevent any disease, injury, or disability. Please consult your healthcare provider for any medical concerns.